Magnetism: An unexpected push for the hydrogen economy

Humankind has entered uncharted territory: atmospheric CO2 levels have soared to a record-breaking 415 ppm for the first time in human history. The need to find a sustainable alternative to CO2-producing fuels is urgent. One of the most promising and environmentally friendly energetic sources is hydrogen generated via water splitting, the reaction in which water is broken down into oxygen and hydrogen. Now, researchers from the Institute of Chemical Research of Catalonia are bringing this hydrogen economy one step closer in an unexpected way.
In a paper published in Nature Energy, scientists from the groups of Galán-Mascarós and López report using a magnet to directly enhance the production of hydrogen in alkaline water splitting via electrolysis. "The simplicity of the discovery opens new opportunities to implement magnetic enhancement in water splitting. Furthermore, the low cost of the technology makes it suitable for industrial applications," says Felipe A. Garcés-Pineda, first author of the paper.
Magnetic pull
The researchers report that the presence of an external magnetic field induced by approaching a neodymium magnet to the electrolyser spurs the electrocatalytic activity on the anode in some cases, increasing the hydrogen production two-fold. The scientists report that the magnetic field directly affects the reaction pathway by allowing for spin conservation of the active catalyst, which in turn favours parallel spin alignment of the oxygen atoms during the reaction. Due to the external magnetic field, this overall spin polarization improves the efficiency of the process. "This demonstrates that there is a lot to learn from the intimate reaction mechanisms taking place on electrocatalysts and opens new ways to overcome the limitations of state-of-the-art systems," says Núria López, ICIQ group leader and co-author of the manuscript.
-

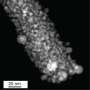
Hydrogen bubbles formed through the water splitting reaction via magnetic enhanceent. Credit: ICIQ -

There's an observable increase in the hydrogen bubble formation when the magnet is approached to the anode. Credit: ICIQ
The researchers studied a variety of catalysts in identical working conditions and report that the catalytic activity enhancement is proportional to the magnetic nature of the catalysts used to drive the water-splitting reaction. Thus, NiZnFe4Ox, a highly magnetic ferrite, exhibited the greatest enhancing effect when presented with a magnetic field. This ferrite can also magnetically attach itself to a nickel metal support, curbing the need to use binders to attach catalysts to a physical support.
Big science for big problems
"The challenge for a hydrogen economy is not only a scientific one," explains José Ramón Galán-Mascarós, ICIQ group leader and corresponding author of the paper. He says finding technological solutions that avoid the use of noble metals, such as platinum or iridium, is the real challenge. It's also necessary to make the hydrogen energy cycle viable. Since noble metals are expensive and extremely scarce, their use limits the scaling-up of the technologies for mass production. Instead, scientists are searching for earth-abundant alternatives, which offer very good performance in alkaline conditions and allow for economically viable scaling.
"After decades of scientific research, the problem is still ongoing and big enough as not to expect easy solutions. The challenge of making sustainable fuels requires a multidisciplinary effort, and ultimately, international collaboration," concludes Galán Mascarós.
More information: Felipe A. Garcés-Pineda, Marta Blasco-Ahicart, David Nieto-Castro, Núria López and José Ramón Galán-Mascarós. Direct magnetic enhancement of electrocatalytic water oxidation in alkaline media. Nature Energy. 2019. DOI: 10.1038/s41560-019-0404-4